Production at CANDOR
First class reagents are not the result of good fortune!
The production facility of CANDOR Bioscience:
The solutions manufactured by CANDOR Bioscience fulfil the highest demands for quality and low batch-to-batch variations. A very well trained, experienced and highly motivated team is needed to reliably fulfil such high expectations. Our team has to deal with production, process control and batch release analytics in a consistent manner. To enable our team to always provide the highest performance, we invested in the best available equipment and machinery for production. By doing this our team can easily give you premium solutions, every day, every week, every month, year-in-year-out. In our industry sector CANDOR sets up new standards for quality and batch-to-batch consistency. Even our customers who require custom solutions benefit from this modern facility.
Important requirement for our production equipment include:
- Easy-to-operate devices to ensure error-free handling even if there is pressure in the timelines for production.
- Easy-to-clean devices to ensure highest hygienic standards in production.
- Any relevant working step and process control has to be documented. Our documentation has to be traceable in all aspects and without any gaps before, during and after production. This is constituted in SOP’s (standard operating procedures) for production.
- Measurement results of any control devices have to be automatically saved and printed to ensure completeness and integrity of all data-sets for any production batch.
- Worker protection and good ergonomics have to be supported by all of our devices.
In addition to our professional team, our production Know-How and selected high quality raw materials, additionally our production equipment plays an important role in ensuring highest quality for our products.
Part of such equipment will be presented here to demonstrate what our philosophy is. Quality is not just a word for us. Quality is our philosophy. We are certified for DIN EN ISO 9001:2008 and our facility is audited twice-yearly or inspected according to the rules of this certification. Additionally customers may always ask for personal audits in our facility. Let’s have a look at some of the devices, which enable us to be the quality-leader in our industry.
Water Purification System – TKA

Each of the solutions and buffers of CANDOR have one major component: Water! It is obvious that we have to focus on the highest purity of our water for production. Not to mention that we cannot accept changes in water quality during production of our solutions. Thus we checked many different kinds of water purification systems and different suppliers. Our choice is to work with a water purification system produced by TKA (Part of Thermo Fisher Scientific). This system has been set up and is under the constant control of our own technicians as well as periodical maintenance by TKA service technicians. This gives us the safety to have high quality water for production at any time. The first stage of expansion of our system, which has been set up, has a daily production capacity of 2,500 litres. Additional identical modules can be implemented to replicate this capacity without changing the process of purification and thus without changing the water quality. Important parameters of water quality like conductivity or TOC are continuously monitored. Control limits are implemented and automatically checked. Control measurements are documented regularly.
Please find additional information here.
Magnetic stirrer system – 2mag

CANDOR produces buffer solutions in industrial sizes. Thus we have to mix salts, buffer components, proteins, organic and inorganic raw materials and other biomolecules in all batch sizes. Standard batches at our facility range from 1 litre to 1000 litres. Higher volumes are available on request. It is obvious that we need a very good mixing system, which not only works reliably with consistent and adjustable forces, but with high speed and very effectively. We need a system, which has nearly the same characteristics starting from 1 litre pilot-batches up to 300 litres or 1000 litres production batches. Up-scaling is a major task for industrial production of modern solutions. We cannot accept changes in mixing efficiencies, speed or characteristics during scale-up processes. Thus we are equipped with the complete family of magnetic stirrer products from 2mag AG. We can use the same mixing technology for any volume and can adjust mixing forces and speed product-specifically. Production paramaters for mixing can be fixed in our SOP’s and at any of our different production stations, so that we can run the same mixing program for the same product. This adds reliability and robustness to our production processes. Furthermore the 2mag drives are made for permanent operation and thus we can run them day-in-day-out for any period needed. The surfaces of our magnetic stirrer have tight stainless steel housing. Cleaning procedures can be done easily for maximum hygienic standards in all our production runs.
Process control like conductivity and pH-values – SI Analytics
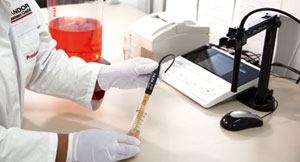
Important aspects for process parameter control devices are:
Highly reproducible measurements in any kind of solution (ranging from high purity water up to highly concentrated protein solutions).
- Maximum independence of the results from process parameters like temperature changes in the solution.
- Easy documentation with data-integrity.
- Appropriate user interface for easy and fast measurements.
We decided to use very robust production control devices at each production station in addition to a central batch-release testing system (ProLab 4000) which meets FDA standards for documentation and data-integrity. Iodine/Iodide reference system electrodes are used for pH-measurements. This enables fast response times, very good robustness of the electrodes as well as useability of the same electrode type for protein-free and proteinaceous solutions. Furthermore this modern reference system is less sensitive to temperature changes than other ones, e.g. as Ag/AgCl.
Please find additional information here.
Weighing according to FDA standards – Mettler-Toledo

Weighing processes are part of every production process at CANDOR. Thus we have to rely completely on the accuracy of our weighing. Starting from the analytical balances and precision balances up to industrial weighing platforms with adequate terminals we chose the high-end products from Mettler-Toledo. All such instruments meet FDA requirements for analytical precision, reproducibility and documentation procedures. Furthermore they are easy-to-operate, the terminals are easy-to-read and they are optimized for easy cleaning. The industrial platforms and terminals are completely covered with stainless steel for easy-cleaning and highest hygienic standards. All scales are monitored by our own technicians and additionally we run service contracts with Mettler-Toledo for all these instruments. Thus every scale is maintained and inspected routinely. Furthermore the calibration service of Mettler-Toledo ensures correctness and traceability of results at any time.
Laboratory furniture – Waldner’s Scala system
Even the laboratory furniture is an important detail for the facility. We chose the Scala system of Waldner to have modern and ergonomically optimized workplaces. Waldner is the market leader in lab furniture and the new system is designed for optimum ergonomics as well as highest flexibility. Our team is proud of individually planned workplaces, which can even be adapted to future needs. Our team was able to discuss all our requirements with the Waldner construction team and after this CANDOR was able to get an adequate and well-suited system – specific to our needs.






